Henrik Nakskov
Dir, Partner
Phone: +45 31 33 37 42
[email protected]
LinkedIn
CIMS works from the CenterPoint of data, information and processes in drug development. Our services are centred around data where we can support specialist, in-depth and cross functions as needed. We facilitate that data and processes are integrated and in regulatory compliance.
CIMS provides both consulting assistance as well as development and implementation of specific solutions.
CIMS focus is a holistic advice under Information Management where the entire value chain of information management, IT, QMS and the organisation is taken into account.
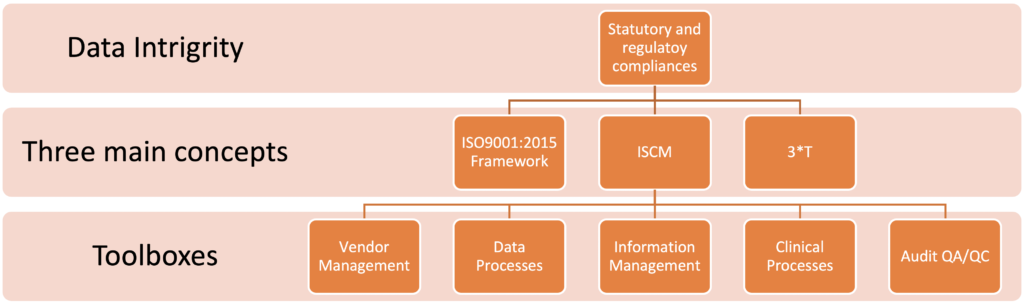
When CIMS is working with data integrity the main concepts are ISO 9001:2015 inspired risk-based thinking, Information Supply Chain Management (ISCM) and 3*T perspective (Truth-Trust-Traceability). Together these three main concepts hold the key to successful data integrity.

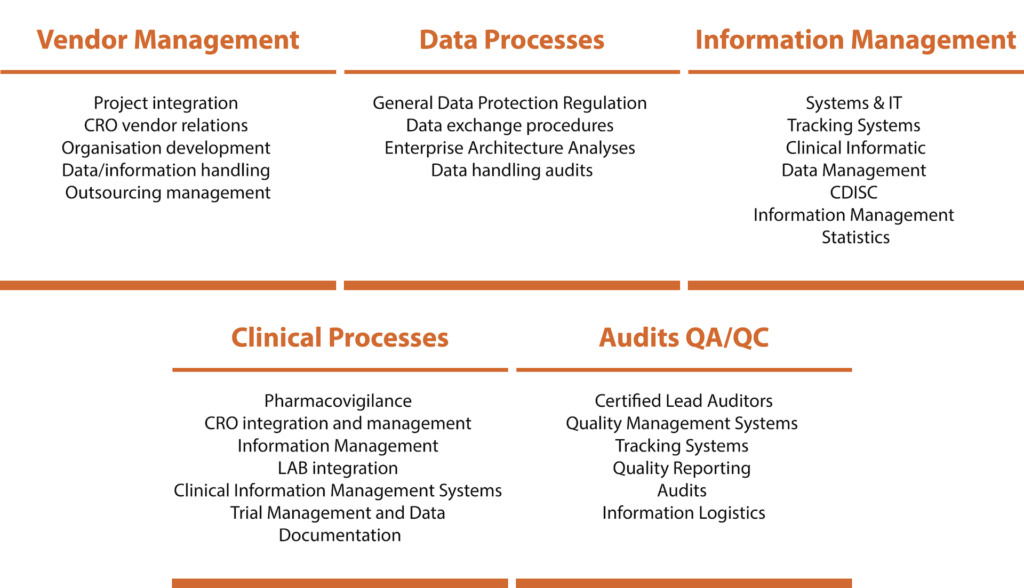
For each main concept we leverage five toolboxes of the data exchanges and processes.
The Toolboxes we use are multifaceted for ensuring compliances with statutory and regulatory requirements.
Bringing it all together the statutory and regulatory compliance is secured by combining CIMS three main concepts and using the appropriate tools from our toolboxes.
Setting Data Integrity as a goal gives you as our customer a new insight into processes which leads to a very efficient oversight of all integrated activities and deliverables.
With our integrated toolbox and three concepts CIMS can monitor your data integrity.
There are always challenges and they can be handled.
Wants to know more – Get a free hour talk with our senior consultants!
Here are some of our customers statements describing the difference CIMS has made for them.
Beside securing data integrity we also deliver stand alone services like shown below. Please ask us for specific tasks not mentioned here.
Decisions based upon good data integrity will multiply the value of your information significantly. We help you to use the growing wave of data to your advantages, increasing your company’s revenue years ahead!
It’s not only about technology. It’s about your processes, information management, staff and organisation using technology and data to your own favor.
We are bridging the gap between data growth, information management, processes, security and quality all the way in the information supply chain.
CIMS can help you with all the challenges of Information Management in Clinical Drug Development.


CIMS Denmark
Strandvejen 301, 2.
2920 Charlottenlund
Denmark
CIMS India
#17, Patel Ram Reddy Road,
Krishna Reddy Layout,
Domlur, Bangalore 560071
India
Henrik Nakskov
+45 31 33 37 42
Email: [email protected]